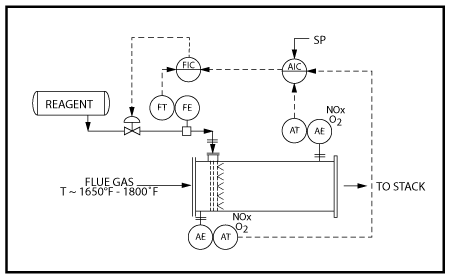
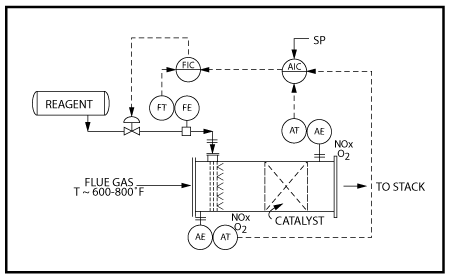
PCC puts our proprietary technology and combustion processes know-how to work selecting the optimum burner(s), system configuration, process design, and operating conditions to meet the specified NOx emission limits. All NOx generation methods – “thermal NOx formation”, “chemical NOx formation” and “prompt NOx formation” are evaluated when determining whether low NOx burners, staged air systems, staged fuel systems and/or multi-stage combustion systems are required to achieve low NOx emissions.
What our customers are saying...
"PCC... The best-kept secret in the industry."
— Rohm & Haas
"PCC works like a well-oiled machine."
— Louisiana Pigments Company
"We would not be where we are today without the engineering knowledge and efforts, quality of construction, professionalism, and cooperation of your first-class organization."
— Montauk Energy Capital
"PCC's commitment to safety and quality allow completion ahead of schedule, under budget, and safely."
—
"PCC's desire to deliver a quality product was apparent throughout all phases of our project, and PCC's overall gas combustion experience resulted in a robust and reliable operating unit."
—
"We are very pleased that it was commissioned five weeks earlier than the contract completion date."
—
"PCC was absolutely on time with delivery of our thermal oxidizer... I was quite impressed. All the units we bought from PCC run flawlessly, even after 10 years. We can't calculate a MTBF, because there have been no failures."
—